Kim-Anne Lê, PhD
Department of Physiology, University of Lausanne and Service of Endocrinology, Diabetes and Metabolism, Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland; Childhood Obesity Research Center, Department of Preventive Medicine, University of Southern California, Los Angeles, CA, USA
luc.tappy@unil.ch

Key Points
- Added sugars account for the major part of dietary fructose.
- Fructose is nearly completely converted into triose phosphate in the liver. Triose phosphates are secondarily converted into glucose and glycogen, lactate, and to a lesser extent, fatty acids; fructose is a potent lipogenic agent in the liver.
- In rodents, high-sucrose or high-fructose diet has been associated with obesity, diabetes mellitus, dyslipidemia, and high blood pressure.
- In humans, fructose overfeeding causes mild hepatic insulin resistance, hyperlipidemia, ectopic lipid deposition in muscle fibres and liver cells, and increases intra-abdominal (visceral) adipose tissue.
- The metabolic effects of free fructose, as in high-fructose corn syrup and of bound fructose as in the sucrose moiety, appear essentially the same.
- There is overwhelming evidence that an overconsumption of sucrose or of fructose can produce deleterious metabolic effects.
Sucrose, fructose and glucose are caloric sweeteners. Fructose and glucose are monosaccharides present in small amounts in fruits and honey, while sucrose is found in substantial amounts in sugar cane and beets. Sucrose and fructose are not essential components of Mans feeding, and their consumption has remained low throughout the prehistory and middle age. Sucrose consumption in Europe increased essentially during the nineteenth century, and presently represents 10-25% of total energy intake in most parts of the world. Sweetened beverages are major contributors to sugar intake, and represent up to 10% total energy in North America [1-2].
It has long been noticed that high-sugar intake may have adverse health effects. In rodents, consumption of a high-sucrose diet leads to the development of obesity, insulin resistance, diabetes, dyslipidemia, fatty liver, and high blood pressure [3]. More than 50 years ago, it had already been suspected that consumption of refined sugar in humans may be linked to dyslipidemia and coronary heart disease [4-5].
The sucrose moiety is constituted by one molecule of glucose linked to one molecule of fructose. In the gut, sucrose is split into glucose and fructose before being absorbed into the portal blood. Fructose and glucose thereafter have a quite different metabolic fate. The initial steps of fructose metabolism do not require insulin. Due to this non-dependency upon insulin secretion, and to the fact that fructose ingestion does not markedly increase glycemia, fructose has once been thought to be a suitable sweetener for diabetic patients. After glucose ingestion, a minor (≈15%) portion of the absorbed glucose is extracted by the liver through the transporter GLUT2. Once inside the cell, glucose is converted into glucose-6-phosphate by the enzyme glucokinase, the expression of which is dependent on insulin. Glucose-6-phosphate is then converted into pyruvate in the glycolysis pathway. Key glycolytic enzymes are inhibited by citrate and ATP, and hence hepatic glucose degradation is modulated according to the energy status of liver cells. After fructose ingestion, fructose, like glucose, is transported into hepatic cells by GLUT2. Once inside the cells, it is rapidly converted to fructose-1-phosphate and to glyceraldehyde-phosphate and dihydroxy-acetone-phosphate by specific enzymes (fructokinase and aldolase B). In contrast to glycolysis, these initial steps for fructose degradation are not regulated by the energy status of the cell, and hence the near total amount of fructose reaching the liver is metabolized into triose phosphates, which are secondarily converted into glucose, glycogen, lactate, and, to a smaller extent, lipids. Due to this large hepatic synthesis of triose phosphates, which are lipogenic precursors, fructose is probably the most potent lipogenic nutrient in our diet [6-7].
There is strong evidence, both in animal models and in humans, that a hyperenergetic high-sucrose diet produces adverse metabolic effects, of which an increase in plasma triglycerides is the most striking. This hyperlipemic effect appears to be even enhanced in overweight, insulin-resistant patients [8]. There is strong evidence that the fructose component of sucrose is essentially responsible for these effects. Fructose administration acutely impairs hepatic insulin actions in healthy human subjects [9]. Consumption of a hyperenergetic high-fructose diet also rapidly produces a slight, but significant increase in fasting plasma glucose and in hepatic glucose production, indicating some degree of hepatic insulin resistance [10].
An increased dietary fructose intake rapidly and consistently raises fasting plasma triglycerides. This effect is observed in both healthy, normoglycemic subjects and in patients with type 2 diabetes [11-12]. There is evidence that a stimulation of hepatic de novo lipogenesis plays an prominent role in this process since: 1) fructose markedly increases fractional hepatic de novo lipogenesis; 2) the increase in fasting plasma triglycerides correlates with the increase in hepatic de novo lipogenesis; and 3) dietary supplementation with n-3 fatty acids, which downregulate de novo lipogenesis, reduces both de novo lipogenesis and fasting plasma triglycerides [13]. Besides a significant increase in fasting plasma triglyceride concentrations, a high-fructose intake also enhances postprandial lipemia, due, at least in part, to a decreased clearance of triglyceride-rich lipoproteins [13]. In overweight subjects, dietary supplementation with fructose-sweetened drinks increased body weight, hepatic de novo lipogenesis, and intra-abdominal (visceral) fat volume, and impaired glucose tolerance. In contrast, administration of glucose-sweetened drinks did not produce these effects. Total body weight gain was, however, similar with fructose and glucose drinks [14].
In view of the rapid effects of fructose on plasma lipids and on hepatic insulin sensitivity, it was surprising to observe that, in healthy, insulin-sensitive subjects, hyperenergetic high-fructose diets, administered for as long as 4 weeks, failed to decrease whole body insulin sensitivity, suggesting that skeletal muscle (which is the major contributor to whole body insulin sensitivity) was not directly affected by fructose.
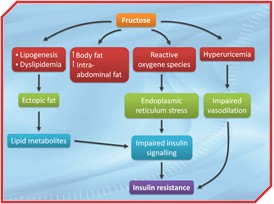 [Click to enlarge]
[Click to enlarge]
There are, however, several mechanisms by which fructose may in the long term induce insulin resistance (Figure). Short-term fructose administration significantly increased intrahepatic and intramuscular lipid concentrations. Since ectopic lipid deposition appears to be associated to insulin resistance, this observation raises the concern that longer-term effects of fructose administration may be associated with significant lipotoxicity, and may eventually lead to more generalized insulin resistance. There is also the concern that fructose intake may contribute to the development of non-alcoholic fatty liver disease. Furthermore, a high sucrose or fructose intake, when associated with a hyperenergetic diet, is expected to increase body weight and body fat mass, which may secondarily cause insulin resistance [15-16]. Besides a stimulation of de novo lipogenesis and lipotoxicity, other mechanisms may also be involved in fructose-induced insulin resistance. Fructose administration produces an oxidative stress through the generation of reactive oxygen species, and triggers an endoplasmic reticulum stress response, which may be associated with impaired insulin signalling [17]. In addition, fructose increases uric acid production due to its rapid and complete phosphorylation to fructose-1-phopshate, which produces massive hepatic degradation of ATP to ADP and AMP. The ensuing hyperuricemia may cause an endothelial cell dysfunction, resulting in an impaired postprandial muscle vasodilation, and this phenomenon may contribute to insulin resistance [18].
There has been much debate over the past years regarding the role of dietary high-fructose corn syrup (HFCS) in the pathogenesis of obesity and of metabolic diseases at large. Since the 1960s the corn industry developed technologies to extract starch from corn, hydrolyze it to glucose, and convert part of the glucose into fructose through enzymatic isomerisation. This resulted in the production of HFCS. HFCS can be produced using various fructose:glucose ratio, the most commonly used being HFCS-55, containing 55% fructose and 45% glucose, i.e. a fructose:glucose ratio close to the 1:1 ratio found in sucrose. Due to the high sweetening power of HFCS, together with its low production cost, HFCS utilization rapidly increased in North America over the past 50 years, and HFCS has replaced sucrose in a substantial portion of sweetened beverages. Although the increase in HFCS consumption roughly parallels the increase in obesity prevalence in the US, however to date there has been no solid evidence that the consumption of HFCS-55 produces different metabolic effects than equivalent amounts of sucrose [19]. However, this point remains unsettled, and the results of several ongoing studies are awaited.
Based on the data presently available, there is overwhelming evidence that an overconsumption of sucrose or of fructose can produce deleterious metabolic effects. Fructose can indeed increase fasting and prostprandial lipemia, can produce a mild impairment of hepatic insulin sensitivity, and can increase ectopic fat depots. Hypercaloric fructose intake will also increase total body fat and intra-abdominal fat mass, which may in turn contribute to insulin resistance and metabolic disorders. Initiatives aimed at reducing sugar intake are therefore likely to reduce overweight and metabolic disorders. However, there is a number of issues which presently remain unsettled:
- Most studies, which have addressed the potential mechanisms by which fructose may contribute to metabolic diseases, have used large fructose intake together with high energy intake. There is an urgent need evaluating the effects of sucrose and fructose intake as part of an isoenergetic diet.
- Athletes are a special group of subjects, with very high physical activity and energy expenditure. These subjects often consume a very high amount of sugars, but are not at risk of (and even appear to some extent protected against) metabolic diseases. How physical activity modulates the effects of fructose, and whether physical activity modifies the maximal "safe" intake of sugars need to be determined.
- The metabolic responses to fructose appear to be blunted in premenopausal women [19-20]. However, overweight women have similar risk of developing metabolic diseases such as diabetes than overweight men. More data are needed regarding the health effects of sugar according to sex.
- A limited amount of data suggests that the deleterious effects of sugars may be enhanced in specific subgroups of individuals (offspring of patients with type 2 diabetes, overweight, insulin-resistant subjects). This may have practical implications regarding personalized dietary recommendations.
- There is ample evidence that a hyperenergetic, high-sucrose diet produces adverse metabolic effects in humans. There is also epidemiological evidence showing that obesity and metabolic diseases are associated with high-sugar intake and/or high sweetened beverages intake. Intervention studies are however still needed to demonstrate that a reduction of sugar intake indeed decreases cardiometabolic risk factors in patients with metabolic disorders.
References
- Vos MB, Kimmons JE, Gillespie C, et al. Dietary fructose consumption among US children and adults: the Third National Health and Nutrition Examination Survey. Medscape J Med 2008; 10: 160.
- Malik VS, Popkin BM, Bray GA, et al. Sugar-sweetened beverages, obesity, type 2 diabetes mellitus, and cardiovascular disease risk. Circulation 2010; 121: 1356-64.
- Bizeau ME and Pagliassotti MJ. Hepatic adaptations to sucrose and fructose. Metabolism 2005; 54: 1189-201.
- Yudkin J and Roddy J. Levels of dietary sucrose in patients with occlusive atherosclerotic disease. Lancet 1964; 2: 6-8.
- Bantle JP, Laine DC and Thomas JW. Metabolic effects of dietary fructose and sucrose in types I and II diabetic subjects. JAMA 1986; 256: 3241-6.
- Mayes PA. Intermediary metabolism of fructose. Am J Clin Nutr 1993; 58: 754S-65S.
- Tappy L and Le KA. Metabolic effects of fructose and the worldwide increase in obesity. Physiol Rev 2010; 90: 23-46.
- Minehira K, Vega N, Vidal H, et al. Effect of carbohydrate overfeeding on whole body macronutrient metabolism and expression of lipogenic enzymes in adipose tissue of lean and overweight humans. Int J Obes Relat Metab Disord 2004; 28: 1291-8.
- Dirlewanger M, Schneiter P, Jequier E, et al. Effects of fructose on hepatic glucose metabolism in humans. Am J Physiol Endocrinol Metab 2000; 279: E907-11.
- Faeh D, Minehira K, Schwarz JM, et al. Effect of fructose overfeeding and fish oil administration on hepatic de novo lipogenesis and insulin sensitivity in healthy men. Diabetes 2005; 54: 1907-13.
- Bantle JP, Raatz SK, Thomas W, et al. Effects of dietary fructose on plasma lipids in healthy subjects. Am J Clin Nutr 2000; 72: 1128-34.
- Bantle JP, Swanson JE, Thomas W, et al. Metabolic effects of dietary fructose in diabetic subjects. Diabetes Care 1992; 15: 1468-76.
- Chong MF, Fielding BA and Frayn KN. Mechanisms for the acute effect of fructose on postprandial lipemia. Am J Clin Nutr 2007; 85: 1511-20.
- Stanhope KL, Schwarz JM, Keim NL, et al. Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. J Clin Invest 2009; 119: 1322-34.
- Le KA, Faeh D, Stettler R, et al. A 4-wk high-fructose diet alters lipid metabolism without affecting insulin sensitivity or ectopic lipids in healthy humans. Am J Clin Nutr 2006; 84: 1374-9.
- Le KA, Ith M, Kreis R, et al. Fructose overconsumption causes dyslipidemia and ectopic lipid deposition in healthy subjects with and without a family history of type 2 diabetes. Am J Clin Nutr 2009; 89: 1760-5.
- Wei Y, Wang D and Pagliassotti MJ. Fructose selectively modulates c-jun N-terminal kinase activity and insulin signaling in rat primary hepatocytes. J Nutr 2005; 135: 1642-6.
- Nakagawa T, Hu H, Zharikov S, et al. A causal role for uric acid in fructose-induced metabolic syndrome. Am J Physiol Renal Physiol 2006; 290: F625-31.
- White JS. Straight talk about high-fructose corn syrup: what it is and what it ain't. Am J Clin Nutr 2008; 88: 1716S-21S.
- Tran C, Jacot-Descombes D, Lecoultre V, et al. Sex differences in lipid and glucose kinetics after ingestion of an acute oral fructose load. Br J Nutr 2010; 104: 1139-47.
Download this article (PDF version, 1.1 Mb) Download this issue (PDF version, 3.27 Mb)




